Two hemispheres fit together and pull apart easily until the air is sucked out from between them.
WATCH THE VIDEO
Teachable Topics:
- Atmospheric Pressure
- Vacuums
- Forces
Theory:
Air molecules collide with the surface of an object that's exposed to the air. Many billions of molecules are striking such an object every minute. Even though atoms are really, really small (really), you'd think that so many of them crashing into the object together would exert some kind of force on it. Indeed they do... such a force is called air pressure. The typical pressure of the atmosphere on your skin is 1.01 Newtons for every square centimeter of your body.
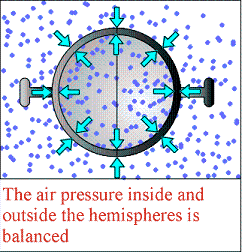
Figure 1: Atmospheric Pressure
Magdeburg hemispheres are two half-spheres of equal size. Placing them together traps air between them. This air is merely trapped, and not compressed, so the pressure inside is the same as the pressure of the atmosphere outside the spheres. That is to say, the air is pushing equally strongly inside as it is outside. The spheres thus pull apart with nearly no resistance.
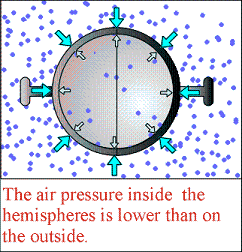
Figure 2: Difference in Air Pressure
Now what if all the air were removed from the inside? Then no air remains to push on the inside of the sphere, but the air outside is pushing at atmospheric pressure. The push from the outside isn't balanced by another push on the inside, so the two hemispheres become very hard to separate.
Apparatus:
- Two spring scales
- Magdeburg Hemispheres
- Vacuum pump

Procedure:
- Place the two Magdeburg hemispheres together tightly and close off the valve.
- Attach one spring scale to each hemisphere, then use the spring scales to pull the hemispheres apart.As you pull, the scales will show the small force you use to pull them apart.
- For the second part to this demonstration, place the two halves together and open the valve. Place the vacuum pump over it, pump the air out of the sphere, then close off the valve again.
- As before, hook the two springs to the hemispheres and pull. This time it will be harder to pull the two halves apart, and the scales will show the greater force of your pull.



