A "reactor" of mousetraps and golf balls provide
an intense demonstration of a runaway nuclear process!
Theory:
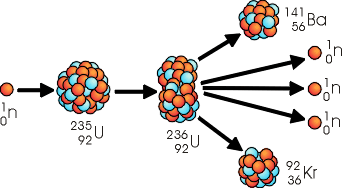
The splitting of a massive nucleus into two fragments, each with a smaller mass than the original is know as nuclear fission. A typical example of nuclear fission is the splitting of a Uranium-235 nucleus. This is a reaction that is used in nuclear reactors to generate heat by which steam is produced and used to turn turbines that generate electricity. The fission of Uranium 235 begins when the Uranium 235 captures a slow moving neutron and forms an unstable "compound nucleus". The compound nucleus quickly disintegrates into a Barium-141 nucleus, a Krypton-92 nucleus, two or three neutrons (2.5 average), and a tremendous amount of energy (~200MeV per fission).
Because the Uranium-235 fission reaction produces 2 or 3 neutrons, it is possible for it to initiate a series of subsequent fission reactions. Each neutron released can initiate another fission event, resulting in the emmission of more neutrons, followed by more fission events, and so on. This is a chain reaction - one event triggers several others, which in turn trigger more events, and so on. In a nuclear power plant the chain reaction is controlled by restricting the number of neutrons available to collide with the Uranium. This is accomplished by absorbing some of the released neutrons with various materials. In an uncontrolled chain reaction (such as an atom bomb explosion) there is nothing to control the number of neutrons being released, so the rate of the chain reaction increases dramatically.
Apparatus:
- 100 modified mousetraps (see "How to Build" section)
- 101 practise golf balls or Ping Pong balls
- A reaction chamber
Procedure:
- Arm the mousetraps and place them in a 10 by 10 grid on the reactor base.
- While positioning the traps, hold them by their sides on the end opposite the trigger paddle and alternate the direction of the trigger paddle (see photo below).
- After positioning a row of mousetraps, place a cardboard partitions around it to avoid triggering armed traps.
- To transport our reactor, we have a 24" by 48" steel cart with large, air-filled tires to reduce bumps while moving the cart.
- We also place a sheet of plywood on top of the cardboard partitions to hold them in place so that if a trap triggers it will only set off one row of traps.
- Below is the reactor ready to be transported to a classroom. Note we don't load the balls on the traps until we are in the classroom.
- Once the reactor is in the classroom, we carefully remove the acrylic cover, the plywood, and the cardboard partitions and we load the balls onto the armed traps.
- Once the acrylic reactor cover is in place, a red coloured trigger ball (representing a neutron) is dropped through a hole in the top of the reactor where it triggers the chain reaction. The whole reaction takes about 2 seconds.
Tips:
- When moving the loaded reactor, use a cart with air-filled tires and move it gently
- Arming the traps is tedious and requires a delicate touch
- Once the reactor is fully set up don't let anyone to near it; the slightest bump could set it off prematurely
- The traps tend to get destroyed after several uses. Try to have extra traps to replace these after performing the demonstration



