The Molecular Motion Demonstrator (MMD) shows how kinetic energy affects how atoms arrange themselves.
WATCH THE VIDEO:
- phase transitions
- solid state physics
- atomic structure
Theory:
The molecules in a solid experience forces from one another. These inter-molecular forces hold many molecules together. To get as close to one another as possible, the molecules form into a highly-ordered structure called a crystal. The molecules in a crystal still have kinetic energy like molecules in the gas phase, but instead of moving around freely, the they remain in their array and vibrate there.
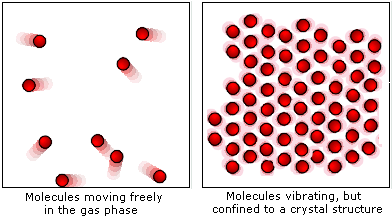
Figure 1: Molecular structure of liquids and solids
The MMD comes with a concave glass dish. Small "molecules" placed in the dish will group near the centre of the dish. This simulates the effect of attractive inter-atomic forces. When the MMD is turned on to a low speed, the molecules jiggle around, but they remain in about the same six-sided "honeycomb" arrangement. This represents molecules at a low temperature in a solid crystalline form.
Increasing the temperature is simulated by turning up the power on the MMD, and this makes the molecules jiggle around more vigorously. If the temperature is further increased, certain molecules begin to slide over one another. Once the molecules can do this, they still clump together, but they no longer stay in a perfect crystalline arrangement and they have more space between one another than before. Such a state represents molecules in the liquid state of matter. Because of the interatomic forces, molecules in a liquid stick together, but because of the higher energy of the molecules, they can move around a little. This freedom to move means that a liquid can easily take the shape of its container.

Figure 2: Molecular structure of solids
Apparatus:
- a concave glass dish
- Molecular Motion Demonstrator
- blue beads*
- one pink bead (or one that stands out from the rest)
Procedure:
- Set the MMD on a flat surface.
- Place the concave plate in the bracket and pour in the beads.
- Turn the MMD on to a low power setting (a 30 degree turn of the knob should give the required speed). The “molecules” jiggle around, but stay in an array like molecules in a solid.
- Increase the speed (by turning the knob about 45 degrees more) to show the molecules as a liquid. The rows of beads should shift and push each other around, but not so much that the beads start to jump about.



